RAM Pharmaceuticals:
Restoring Comfort in Cancer & Dental Care — Through a Patented Dry-Powder Delivery Platform
RAM Pharmaceuticals is advancing a patented dry-powder therapy for severe oral mucositis (Triamdocaine) under FDA IND, and a de-risked 503B dental therapy (Acyclonine MUM). Together, these assets form a platform solution for oral lesion management across oncology and dentistry.
Beyond oncology, our commitment extends to dental health with Acyclonine MUM, a proven therapy providing targeted relief for oral conditions such as mucosal ulcerations, post-operative pain, and traumatic injuries. Through these innovations, RAM Pharmaceuticals is transforming care across two critical health sectors and inviting you to join us on this impactful journey.
Share Price:
$1.00 | Minimum Investment Amount: $1,000.00*
Shares Available: 1,200,000
*A 2% transaction fee applies to all investments.
Select the Opportunity You Want to Review
Both opportunities are part of RAM Pharmaceuticals Inc. and supported by the same patented dry-powder delivery platform.
RAMtherapeutics (Triamdocaine)
Mechanism of Action (Without Disclosing Actives)
Triamdocaine provides:
- A) Rapid Pain Relief
Forms a thin contact layer over exposed nerve endings, reducing nociceptor activation. - B) Local Anti-Inflammatory Effect
Designed to interact at the lesion surface to reduce inflammatory signaling. - C) Protective Micro-Barrier
Powder adhesion shields lesions from mechanical irritation (talking, eating, swallowing).
Actuator Engineering (Highly Relevant to Scientific Investors)
- Dose per actuation: 10.6 mg (uniformity testing underway)
- Particle size distribution optimized for mucosal adherence
- High-velocity dispersion for broad lesion coverage
- 360° rotating head for superior access to:
- Buccal mucosa
- Lingual surfaces
- Tongue underside
- Soft palate
- Posterior oral cavity
- Optional pharyngeal extension for deeper lesions
- Minimal systemic exposure due to localized deposition
Designed for Repeated Use During Cancer Treatment
Regulatory Status
- FDA IND approved
- Phase 2 Proof-of-Concept in preparation
- Endpoints include:
- Change in oral pain scores
- Ability to eat/swallow
- WHO/CTCAE mucositis grade
- Opioid use
- Treatment adherence
- PoC designed to support a request for:
- Combined Phase 2/3 program
- Potential Fast Track designation
This follows a standard oncology development pathway, but more efficiently.
Clinical & Regulatory Strategy
Triamdocaine (IND Program)
- IND active
- Phase 2 PoC design complete
- FDA interaction planned post-PoC for combined Phase 2/3
- Opportunities:
- Fast Track
- Supportive-care designation
- International expansion pathways
Acyclonine MUM (503B Program)
- No FDA drug approval required
- Manufacturing upgrades significantly enhance quality and scalability
- Positioned for national DSO rollout
The Unmet Need In Cancer Care:
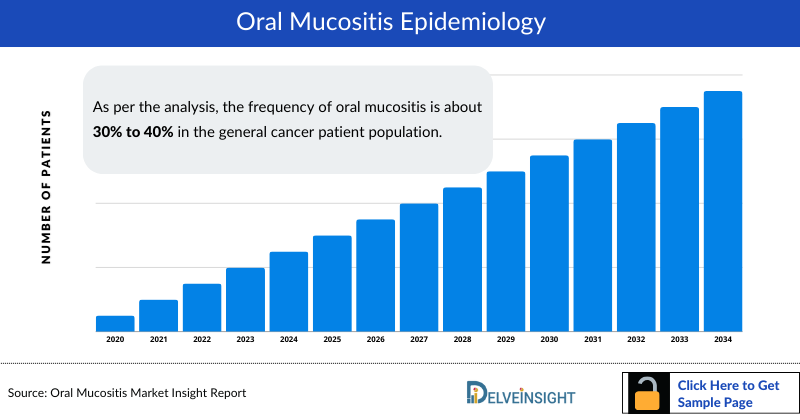
Severe Oral Mucositis (SOM): A Devastating, Treatment-Limiting Complication
- ~40% of chemotherapy patients develop mucositis
- Up to 90% of head & neck chemo/radiation patients
- SOM causes:
- Extreme pain
- Difficulty swallowing
- Inability to eat
- High opioid use
- Feeding tube placement
- Treatment delays
- Hospitalizations
SOM Can Completely Debilitate a Patient
Ulcerations often expose nerve endings, producing constant, opioid-level pain. Patients may lose the ability to:
- Eat
- Swallow
- Speak
- Maintain weight or hydration
Many require:
- Liquid diets
- Feeding tubes
- IV hydration or nutrition
This drastically impacts quality of life and can force oncologists to reduce or interrupt cancer treatment, potentially affecting survival.
Biological Progression of Mucositis
(Scientifically Important)
SOM follows a well-defined five-stage biological cascade:
- Initiation (0–3 days):
Radiation/chemotherapy generate reactive oxygen species → DNA damage. - Signal Amplification (3–6 days):
Inflammatory pathways (TNF-α, IL-1β, NF-κB) trigger epithelial breakdown. - Ulceration (6–12 days):
Deep ulcerations form; nerve endings become exposed → severe pain.
This is the stage Triamdocaine is designed to support. - Secondary Infection (variable):
Bacterial colonization aggravates pain and inflammation. - Healing (12–21 days):
Re-epithelialization occurs if treatment can continue uninterrupted.
Sophisticated investors expect this mechanistic context.
Therapeutic Landscape –
Why Current Options Fail
Existing mucositis therapies include:
- Rinses: Short retention; rapidly diluted in saliva
- Gels: Poor adhesion; limited reach in oral cavity
- Opioids: Systemic side effects; constipation; sedation
- Cryotherapy: Regimen-limited; narrow application
- Palifermin (KGF): High cost; limited indications; IV only
Triamdocaine addresses the core limitations:
- Direct deposition on lesions
- Extended mucosal contact time (vs. rinses)
- Reaches posterior oral cavity
- Repeatable use during every cancer treatment session
- Local action → low systemic exposure
- Consistent dose via actuator
The Cancer Comfort Care Initiative
Pillars of the initiative:
- Direct, rapid comfort for mucosal lesions
- Repeatable use during every radiation or chemotherapy session
- Support for nutrition, hydration, and quality of life
Triamdocaine is the first therapy in this program.
RAMdental (Acyclonine MUM)
Early Commercial Traction and Strong Clinical Pull
Acyclonine MUM, RAM’s 503B dental therapy, demonstrated strong pre-launch demand:
- 250 patients treated in 2 months
- High provider satisfaction
- Rapid word-of-mouth referrals
- Multiple DSOs requesting evaluation and partnership discussions
503B Manufacturing & CMC Upgrades
Your compounder is building a dedicated clean room for MUM, enabling:
- Controlled humidity for powder handling
- Optimized air handling & HEPA particulate control
- Scalable batch manufacturing
- Improved content uniformity
- Stronger quality assurance envelope
Current Status: Uniformity Testing
Uniformity testing is underway to confirm:
- Dose consistency
- Powder distribution homogeneity
- Device-to-device reproducibility
Relaunch Timeline
- Uniformity testing completed: Dec 2025
- Clean room online: Dec 2025
- Commercial relaunch: Late Dec 2025 or early Jan 2026
Market Snapshot
- ~$2.5B estimated global market for oral mucositis and supportive oncology care12
- Defined serviceable segment: radiation- and chemotherapy-induced oral mucositis
- Initial focus: U.S. oncology treatment centers, supported by targeted dental adoption
Oral mucositis is a treatment-limiting complication of cancer therapy.345 Existing options are largely palliative and often constrained by inconsistent delivery to affected tissue.
Market size alone is not the investment thesis. Execution path matters.
Oral mucositis represents a persistent failure within supportive oncology care—not due to lack of therapies, but due to limitations in how therapies are delivered to affected tissue.
Current treatment approaches are largely palliative, with inconsistent lesion contact and limited dwell time, contributing to pain, nutritional compromise, and treatment interruptions.
RAM Pharmaceuticals approaches this market through a targeted delivery platform designed to address this documented bottleneck. Initial focus on U.S. oncology centers allows for disciplined entry, while dental adoption provides real-world validation of the delivery approach and supports future platform expansion.
This strategy reflects a measured, indication-driven path into a large, established market rather than reliance on speculative scale assumptions.
Market estimates derived from published oncology supportive-care literature and industry analyses.
Footnotes
- Global Oral Mucositis Market Size: The global oral mucositis market was estimated at approximately $1.50 billion in 2024 and is projected to reach $2.85 billion by 2033, growing at a CAGR of ~7.35%. Source: Grand View Research, Oral Mucositis Market Size & Share Report.
- Alternative Market Growth Validation: The global oral mucositis treatment market is estimated at $1.99 billion in 2025 and is projected to reach ~$3.72 billion by 2034, driven by rising cancer incidence and supportive care demand. Source: Precedence Research, Oral Mucositis Treatment Market Size, Share & Trends.
- Oral Mucositis Incidence in Cancer Therapy: Approximately 40% of patients treated with chemotherapy develop oral mucositis, with the incidence increasing to ~90% in head and neck cancer patients receiving combined chemotherapy and radiotherapy. Source: Pulito et al., Oral mucositis: the hidden side of cancer therapy, PubMed Central.
- Radiation and Chemoradiation Incidence Rates: Between 30% and 60% of patients receiving radiation therapy develop oral mucositis, and greater than 90% receiving concurrent chemotherapy and radiation are affected. Source: Naidu et al., Chemotherapy-Induced and/or Radiation Therapy-Induced Oral Mucositis, PubMed Central.
- Oral Mucositis as a Supportive-Care Burden: Oral mucositis is a debilitating complication of cancer chemotherapy and radiotherapy, characterized by inflammation, ulceration, pain, and impaired oral function. Source: Colella et al., Interventions for the Prevention of Oral Mucositis in Cancer Patients, PubMed Central.
Traction & Validation
RAMtherapeutics
- IND approved
- Phase 2 endpoints aligned with accepted SOM trials
- Device aligns with real-world lesion locations
RAMdental
- 250 patients treated
- High DSO interest
- Clean room expansion underway
Intellectual Property
Patent Protection Through 2039
IP covers:
- Actuator mechanics
- Powder delivery methods
- Therapeutic application in oral mucositis
- Platform applicability in multiple oral conditions
This creates a broad, defensible moat across both oncology and dental markets.
Use Of Proceeds
Funding will support R&D Development totaling $494,000:
- Phase 2 Proof-of-Concept: $350,000
- IND maintenance & CMC development: $10,000
- RAMdental relaunch & DSO growth: $50,000
- Working capital & IP expansion: $84,000
RAM Pharmaceuticals — Development Roadmap
Key milestones across RAMtherapeutics (Triamdocaine) and RAMdental (Acyclonine MUM).
Offering Snapshot
- Security: Common Stock – RAM Pharmaceuticals Inc.
- Price: $1.00 per share
- Minimum investment: $1,000
- Offering Type: Regulation CF (max $5M)
- Intermediary: DealMaker Securities LLC
Frequently Asked Questions
Regulation CF allows investors to invest in startups and early-growth companies. This is different from helping a company raise money on Kickstarter; with Regulation CF Offerings, you aren’t buying products or merchandise - you are buying a piece of a company and helping it grow.
Accredited investors can invest as much as they want. But if you are NOT an accredited investor, your investment limit depends on either your annual income or net worth, whichever is greater. If the number is less than $124,000, you can only invest 5% of it. If both are greater than $124,000 then your investment limit is 10%.
To calculate your net worth, just add up all of your assets and subtract all of your liabilities (excluding the value of the person’s primary residence). The resulting sum is your net worth.
We cannot give tax advice, and we encourage you to talk with your accountant or tax advisor before making an investment.
Individuals over 18 years of age can invest.
There will always be some risk involved when investing in a startup or small business. And the earlier you get in the more risk that is usually present. If a young company goes out of business, your ownership interest could lose all value. You may have limited voting power to direct the company due to dilution over time. You may also have to wait about five to seven years (if ever) for an exit via acquisition, IPO, etc. Because early-stage companies are still in the process of perfecting their products, services, and business model, nothing is guaranteed. That’s why startups should only be part of a more balanced, overall investment portfolio.
The Common Stock (the "Shares") of Ram Pharmaceuticals (the "Company") are not publicly-traded. As a result, the shares cannot be easily traded or sold. As an investor in a private company, you typically look to receive a return on your investment under the following scenarios: The Company gets acquired by another company. The Company goes public (makes an initial public offering). In those instances, you receive your pro-rata share of the distributions that occur, in the case of acquisition, or you can sell your shares on an exchange. These are both considered long-term exits, taking approximately 5-10 years (and often longer) to see the possibility for an exit. It can sometimes take years to build companies. Sometimes there will not be any return, as a result of business failure.
Shares sold via Regulation Crowdfunding offerings have a one-year lockup period before those shares can be sold under certain conditions. The exceptions are sales to: (i) to the Company; (ii) to an “accredited investor” within the meaning of Rule 501 of Regulation D under the Securities Act; (iii) as part of an offering registered under the Securities Act with the SEC; or (iv) to a member of the Investor’s family or the equivalent, to a trust controlled by the Investor, to a trust created for the benefit of a member of the family of the Investor or equivalent, or in connection with the death or divorce of the Investor or other similar circumstance.
In the event of death, divorce, or similar circumstance, shares can be transferred to: ● The company that issued the securities ● An accredited investor ● A family member (child, stepchild, grandchild, parent, stepparent, grandparent, spouse or equivalent, sibling, mother-in-law, father-in-law, son-in-law, daughter-in-law, brother-in-law, or sister-in-law, including adoptive relationships)
If a company does not reach their minimum funding target, all funds will be returned to the investors after the close of the offering.
All available disclosure information can be found on the offering pages for our Regulation Crowdfunding offering.
You can cancel your investment at any time, for any reason, until 48 hours prior to a closing occurring. If you’ve already funded your investment and your funds are in escrow, your funds will be promptly refunded to you upon cancellation. To submit a request to cancel your investment please email: info@dealmakersecurities.com
At a minimum, the company will be filing with the SEC and posting on it’s website an annual report, along with certified financial statements. Those should be available 120 days after the fiscal year end. If the company meets a reporting exception, or eventually has to file more reported information to the SEC, the reporting described above may end. If these reports end, you may not continually have current financial information about the company.
DealMaker Securities is serving as the intermediary for this offering. Once an offering ends, there is no guarantee that DealMaker Securities will have a relationship with the company. The company may continue its relationship with DealMaker Securities for additional offerings in the future. DealMaker Securities’ affiliates may also provide ongoing services to the company. There is no guarantee any services will continue after the offering ends.